Have you seen these low-grade UTUC tumors?
Left- and right-side
lower-pole tumors
Multifocal
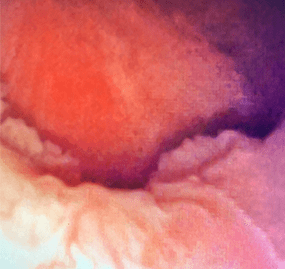
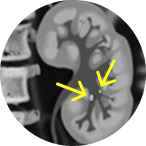
Unresectable, lower-pole
tumor
Unresectable

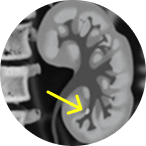
Right mid-ureter tumor
Ureteral
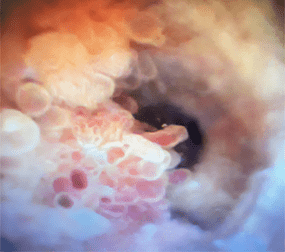
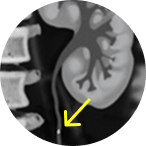
2-3 cm tumor in the
renal pelvis
Large
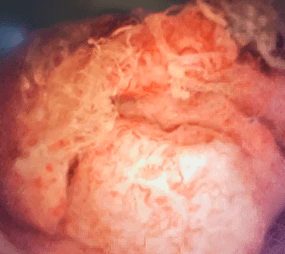
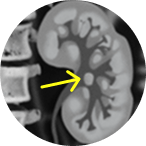
This portrays real-world scenarios in which JELMYTO was used in a diverse range of tumor types. UroGen's Olympus trial studied patients with treatment-naïve or recurrent LG-UTUC with ≥1 measurable papillary tumor 5 to ≤15 mm located above the ureteropelvic junction (tumor debulking was permitted prior to study enrollment for tumors >15 mm).1 For additional inclusion and exclusion criteria, please see the Olympus trial study design.
as your partner
when treating
tumor types like
these
Please click here to see how Jelmyto was studied in the Olympus trial.
Featured resources
Download these resources to learn more and to get your appropriate patients started on Jelmyto treatment.
Indications and Usage
Jelmyto® (mitomycin) for pyelocalyceal solution is indicated for the treatment of adult patients with low-grade Upper Tract Urothelial Cancer (LG-UTUC).
Important Safety Information
Contraindications
Jelmyto® (mitomycin) for pyelocalyceal solution is contraindicated in patients with perforation of the bladder or upper urinary tract.
Warnings and Precautions
Ureteric Obstruction
Ureteric obstruction, including ureteral stenosis and hydronephrosis, occurred in patients receiving Jelmyto. Monitor patients for signs and symptoms of ureteric obstruction, including flank pain, and fever, and for changes in renal function. Patients who experience obstruction may require transient or long-term ureteral stents or alternative procedures. Withhold or permanently discontinue JELMYTO based on the severity of ureteric obstruction.
Bone Marrow Suppression
The use of Jelmyto can result in bone marrow suppression, particularly thrombocytopenia and neutropenia. The following tests should be obtained prior to each treatment: Platelet count, white blood cell count differential and hemoglobin. Withhold JELMYTO for Grade 2 thrombocytopenia or neutropenia. Permanently discontinue for Grade 3 or greater thrombocytopenia or neutropenia.
Embryo-Fetal Toxicity
Based on findings in animals and mechanism of action, Jelmyto can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, administration of mitomycin resulted in teratogenicity. Advise females of reproductive potential to use effective contraception during treatment with JELMYTO and for 6 months following the last dose. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with JELMYTO and for 3 months following the last dose.
Adverse Reactions
Common Adverse Reactions
The most common adverse reactions in ≥ 20% of patients treated with Jelmyto were ureteric obstruction, flank pain, urinary tract infection, hematuria, renal dysfunction, nausea, abdominal pain, fatigue, dysuria, and vomiting.
Additional Adverse Reactions Information
Selected clinically relevant adverse reactions in < 10% and ≥ 2% of patients who received Jelmyto include urinary tract inflammation, bladder spasm, urosepsis, hypersensitivity, and instillation site pain.
Use in Specific Populations
Lactation
Because of the potential for serious adverse reactions in a breastfed child, advise women not to breastfeed during treatment with Jelmyto and for 1 week following the last dose.
Preparation and Administration Information
Jelmyto is for pyelocalyceal use only and not for intravenous use, topical use, or oral administration. Jelmyto must be prepared and administered by a healthcare provider. To ensure proper dosing, it is important to follow the preparation instructions found in the JELMYTO Instructions for Pharmacy and administration instructions found in the JELMYTO Instructions for Administration.
Jelmyto may discolor urine to a violet to blue color following the instillation procedure. Advise patients to avoid contact with urine for at least six hours post-instillation, to void urine sitting on a toilet, and to flush the toilet several times after use.
Jelmyto is a hazardous drug. Follow applicable special handling and disposal procedures.
Please click here for Full Prescribing Information.
Reference: 1. JELMYTO [package insert]. Princeton, NJ: UroGen Pharma, Inc.; 2025.



