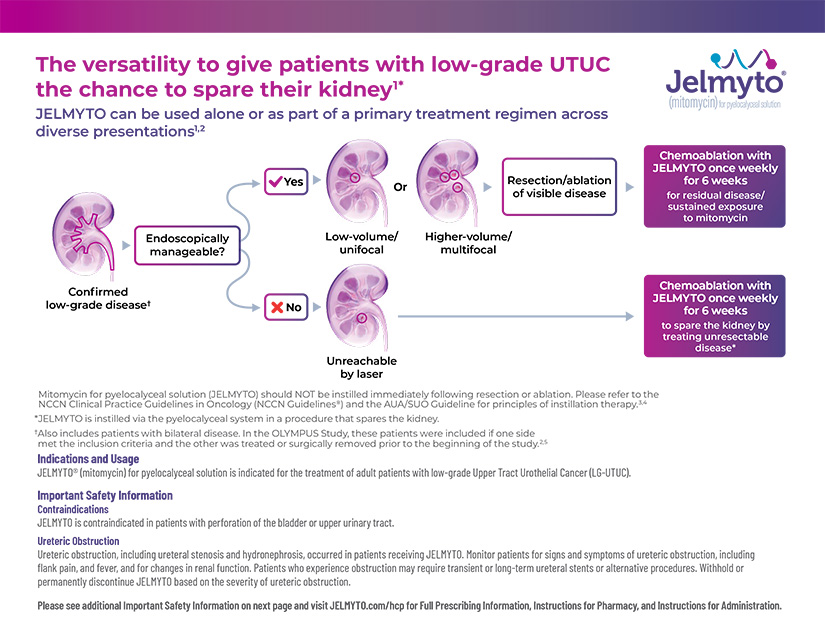
Mitomycin for pyelocalyceal solution (Jelmyto) is now included in both the AUA/SUO Guideline for the Diagnosis and Management of Non-metastatic UTUC and the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®)
The AUA/SUO Guideline for non-metastatic UTUC recognizes Jelmyto as part of a kidney-sparing approach to LG-UTUC management1
recommended
The AUA/SUO Guideline recommends tumor ablation for the initial management of low-risk* favorable† UTUC (Strong Recommendation‡; Evidence Level: Grade B§). In certain clinical situations, chemoablation with Jelmyto® (mitomycin) for pyelocalyceal solution is recommended1
- Location and focality challenges
- Patients for whom age, comorbidities, baseline renal function, and/or procedural risk are considerations
“The advent of new therapies such as reverse thermo-hydrogel preparation of mitomycin have provided an important new means of treating low-risk tumors.”
– Coleman et al1
NCCN Guidelines® for Upper GU Tract Tumors
recommended¶
National Comprehensive Cancer Network® (NCCN®) recommends mitomycin for pyelocalyceal solution (Jelmyto) following complete or near complete endoscopic resection as a primary therapy option for upper tract tumors2#
Most suitably indicated for2
a residual, low-grade, low volume (5-15 mm), solitary tumor in the upper urinary tract
a patient not a candidate for or not seeking nephroureterectomy as a definitive treatment
Mitomycin for pyelocalyceal solution (Jelmyto) may be administered via ureteral catheter or a nephrostomy tube. Complete or near complete endoscopic resection or ablation is recommended prior to mitomycin ureteral gel application.